Beacon Platform — Patient Experience Strategy & IA
Reframing clinical trial participation as a guided decision journey — UX strategy and IA from discovery through enrollment in a regulated research environment.

Lilly's clinical trials platform assumed patient awareness and education within a highly regulated research environment — collapsing complex clinical realities into a linear promotional model that didn't reflect how patients actually navigate a trial decision.
The information architecture treated the enrollment decision like a marketing conversion. Patients needed a guided UX decision framework, not a brochure. The experience gap wasn't informational — it was structural.
My role: I led UX strategy and information architecture for the clinical trial participation platform, working directly with clinical, regulatory, and digital teams. I owned the experience framework, journey design, decision support models, and content hierarchy from discovery through enrollment.
The most important design decision in this project happened before any design work started — reframing what the actual problem was.
"Build a platform that educates patients about Lilly clinical trials and helps them find trials they may be eligible for."
The architecture treats enrollment like a marketing funnel. But patients aren't making a purchase decision — they're making a medical one, under uncertainty, often in a health crisis. The platform needs to be built around how people actually make high-stakes health decisions, not around trial inventory search. The right model is a guided decision journey, not a discovery engine.
This reframe shifted the IA from a search-and-filter taxonomy to a three-stage progressive disclosure model — awareness, consideration, action — structured around patient readiness, not content volume.
I defined a UX framework that mapped what patients needed to understand, feel, and do at each stage before asking them to act. Three stages — awareness, consideration, action — each designed as a standalone UX module that scaled without requiring structural rebuilds.
I owned the IA, content hierarchy, and decision support design across the full platform — translating clinical and regulatory requirements into a patient experience architecture that progressive disclosure logic could hold together across every page.
The phased delivery strategy balanced launch urgency with long-term scalability. Phase 1 established core experience across five sections. Phase 2 expanded on that foundation without requiring architectural rework.
Heuristic review of trial platform IA across pharma and research network sites. AI-augmented pattern extraction across structural models, content hierarchies, and patient pathway design.
All eight platforms surfaced eligibility criteria and enrollment CTAs at the awareness stage. None modeled the cognitive and emotional arc of a patient newly navigating a trial decision.
Stage 1 (Awareness): normalize the concept. Stage 2 (Consideration): evaluate fit in plain language. Stage 3 (Action): eligibility screening and enrollment pathway.
Digital health patient engagement & DTx onboarding. The three-stage model applies to any health platform where users must build understanding before they can meaningfully act — patient recruitment, condition management apps, and health-tech enrollment flows face the identical structural problem.
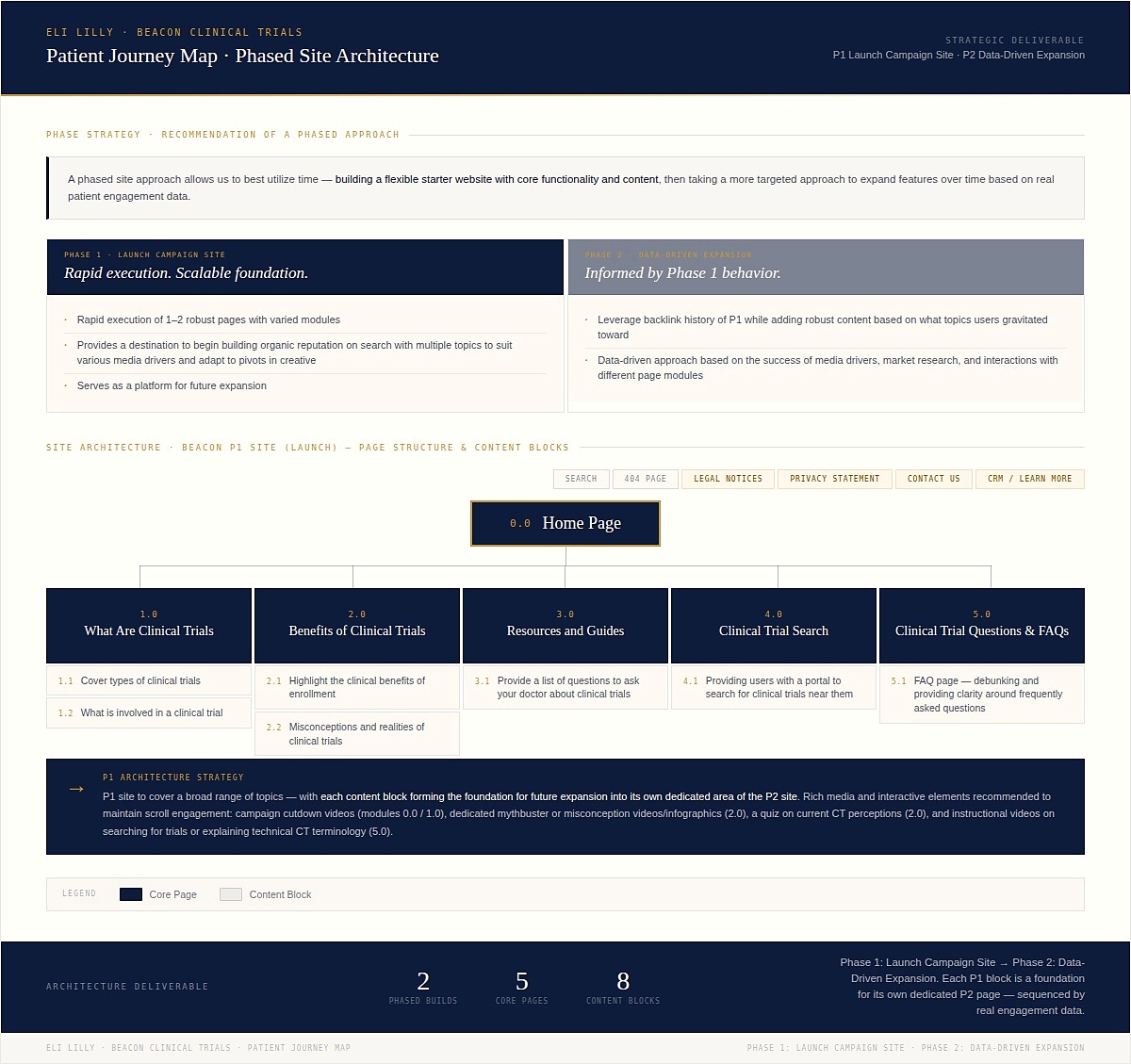
Patient journey map & UX site architecture — phased UX delivery strategy for the Beacon platform, balancing launch urgency with long-term scalability. Phase 1 established core experience across five sections, each designed as a standalone UX module while serving as the foundation for Phase 2 expansion.

Information architecture & experience design framework — translated the UX site architecture into a full information hierarchy model, mapping how clinical content is sequenced across all trial types to support progressive patient understanding from awareness through enrollment.
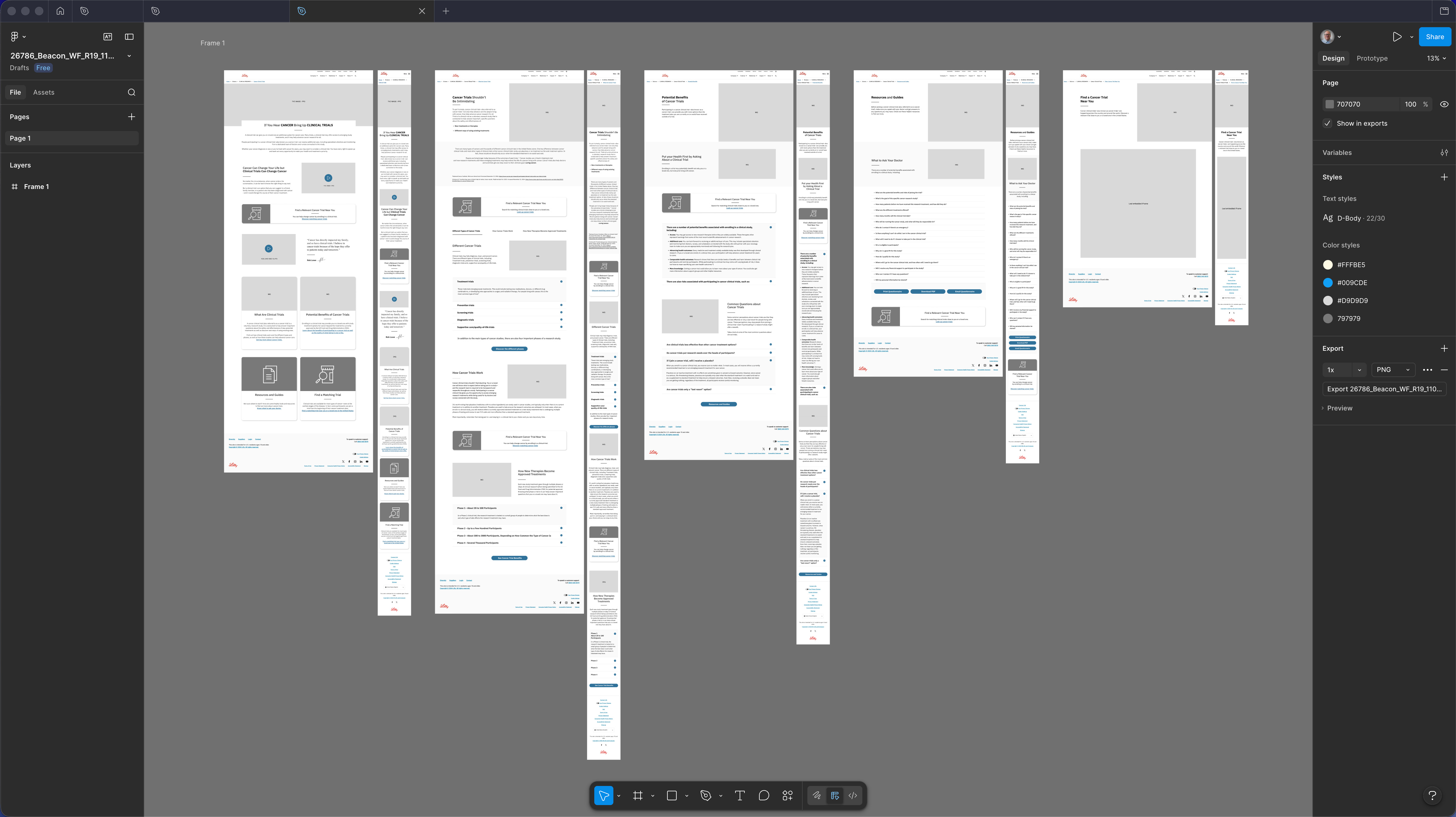
Desktop wireframes R19 — five-section spread showing the complete Beacon patient journey at desktop viewport. Sections in sequence: Homepage (awareness entry, cancer-to-trials narrative hook), Clinical Trials Overview (What Are Trials, Potential Benefits, FAQ accordion), Trial Detail (eligibility criteria in plain-language UX treatment, common questions, risk disclosure integration), Resources & Guides (downloadable decision aids, What to Ask Your Doctor), and Find a Trial Near You (trial-matching interface). Regulatory disclosures designed as structural content components throughout — not footer or modal interrupts. Pre-MLR working copy; structural hierarchy and progressive disclosure logic are the deliverable.
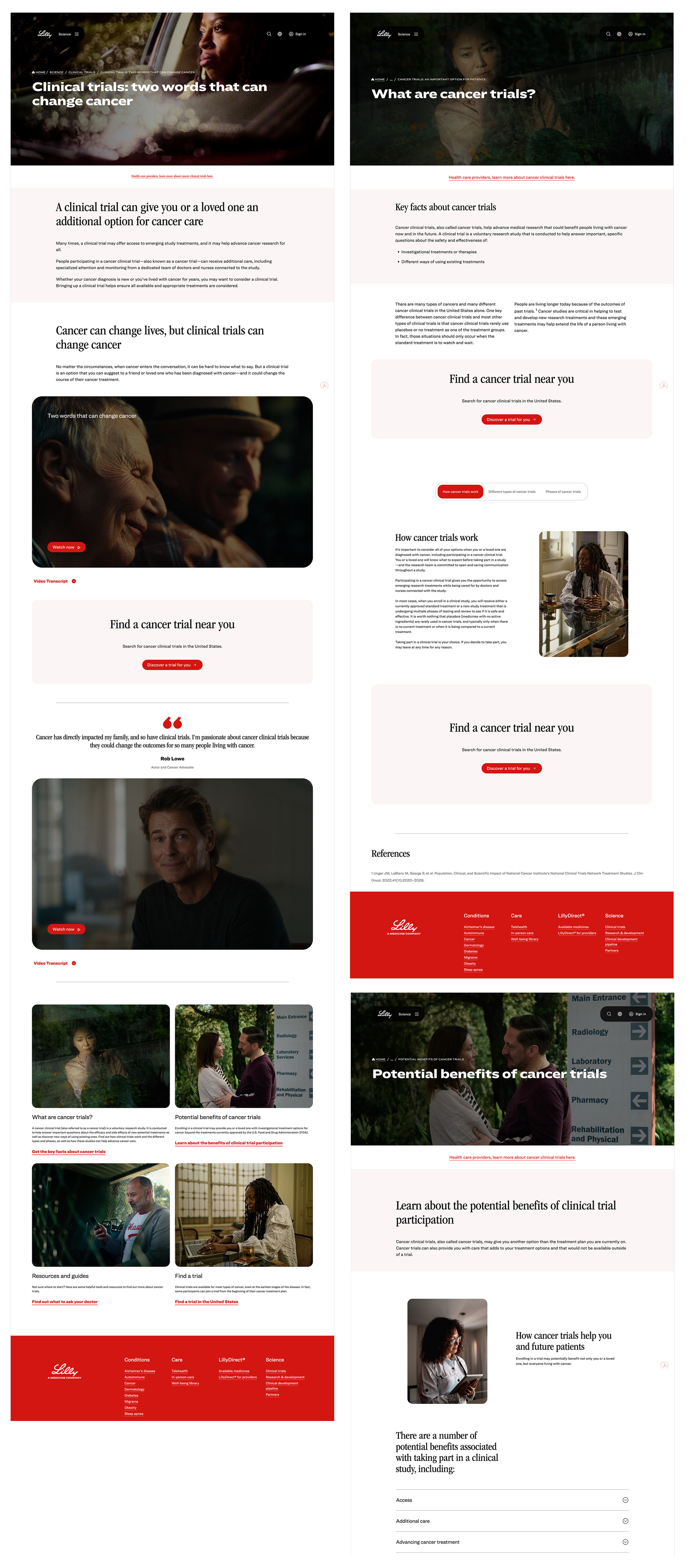
MLR submission artifacts — three representative pages from the complete Beacon platform illustrating UX content hierarchy and progressive disclosure design across three distinct stages of the patient decision journey. Clinical references and regulatory disclosures integrated as structural UX elements.
The progressive disclosure model held across the full platform. Two things I'd refine: first, instrument a readiness signal at entry so patients who arrive already informed — via HCP referral or prior trial experience — can branch at Stage 1 rather than moving through awareness linearly. Second, start the plain-language eligibility negotiation with MLR in the strategy phase, not mid-design. Getting there earlier buys timeline and produces a sharper Stage 2.