Experience Architecture & Competitive Intelligence
Designing oncology experience architecture to support informed treatment understanding across complex, multi-pathway disease management — backed by competitive landscape analysis.

Verzenio serves patients across meaningfully different disease stages — early and metastatic breast cancer — each with distinct treatment goals, support needs, and emotional contexts. A single UX model couldn't serve both without creating confusion or clinical risk.
The experience design gap wasn't informational — it was architectural. Content existed but the UX structure didn't reflect where different patients actually were in their journey. A single undifferentiated model created confusion and clinical risk for EBC patients who didn't yet identify as having a serious disease, and failed MBC patients who needed evidence and context before emotional support.
My role: I led experience design and UX architecture across the Verzenio patient support ecosystem — defining the structural design model that served both patient audiences without requiring separate platforms.
The brief described a content problem. The real issue was a structural one that content alone couldn't solve.
"Redesign the Verzenio DTC platform to better serve both early breast cancer and metastatic breast cancer patients — currently the experience isn't differentiated enough between the two audiences."
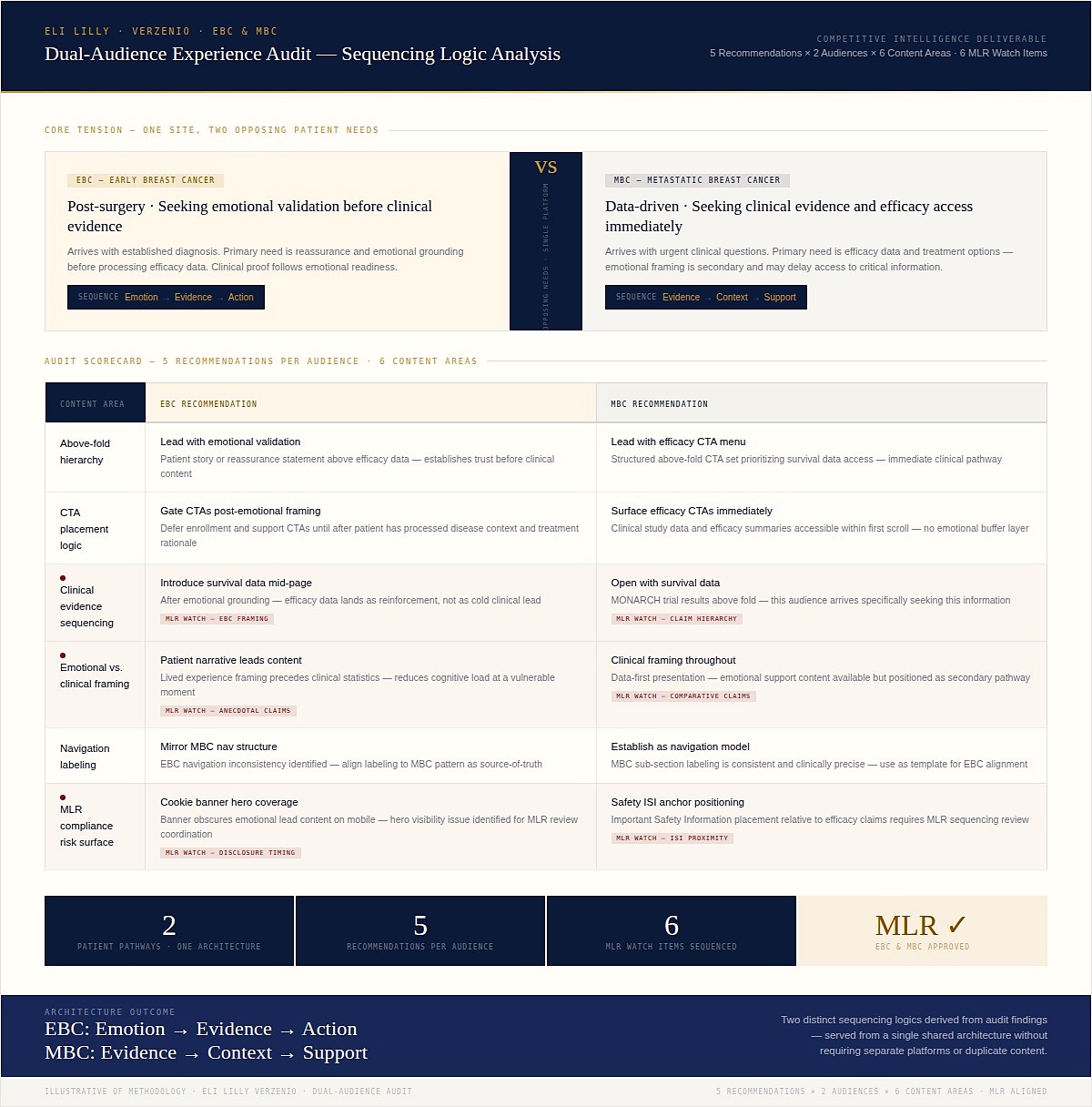
The problem isn't insufficient differentiation — it's that both audiences are running through the same UX sequencing model. EBC and MBC patients don't just need different content; they need fundamentally different information architectures. EBC patients need emotion first — validation before evidence. MBC patients are further along their disease journey and need evidence first — clinical credibility before emotional support. Two sequencing models, not one with variations.
This reframe produced the EBC model (Emotion→Evidence→Action) and the MBC model (Evidence→Context→Support) as structurally distinct sequencing architectures served from one platform infrastructure.
Two UX sequencing models were designed: EBC — Emotion→Evidence→Action; MBC — Evidence→Context→Support. I mapped the full patient experience ecosystem across both disease stages, identifying where UX could share infrastructure and where experience design needed to diverge.
Five UX design recommendations per audience across six content areas, with six MLR watch items sequenced for regulatory review alignment. MBC was identified as the source-of-truth UX navigation pattern, with EBC adapting the structural foundation to its distinct emotional sequencing requirements.
Heuristic evaluation of breast cancer DTC platforms across CDK4/6 inhibitor competitors. AI-augmented structural pattern analysis. EBC vs. MBC disease-stage emotional differences and treatment decision-making research.
Every platform audited applied a single IA across EBC and MBC — efficacy first, then mechanism, then support. No platform accounted for the fundamentally different information needs and emotional contexts between disease stages.
EBC: validate the emotional reality of diagnosis before introducing clinical evidence. MBC: lead with clinical credibility — these patients have been through treatment and evaluate support against a high standard.
Multi-condition health platforms & chronic disease management apps. Designing structurally distinct experience flows for audiences at different disease stages applies to any health platform where user states differ meaningfully — diabetes management, mental health, and chronic condition apps often collapse these differences into one model, creating the same structural failure.
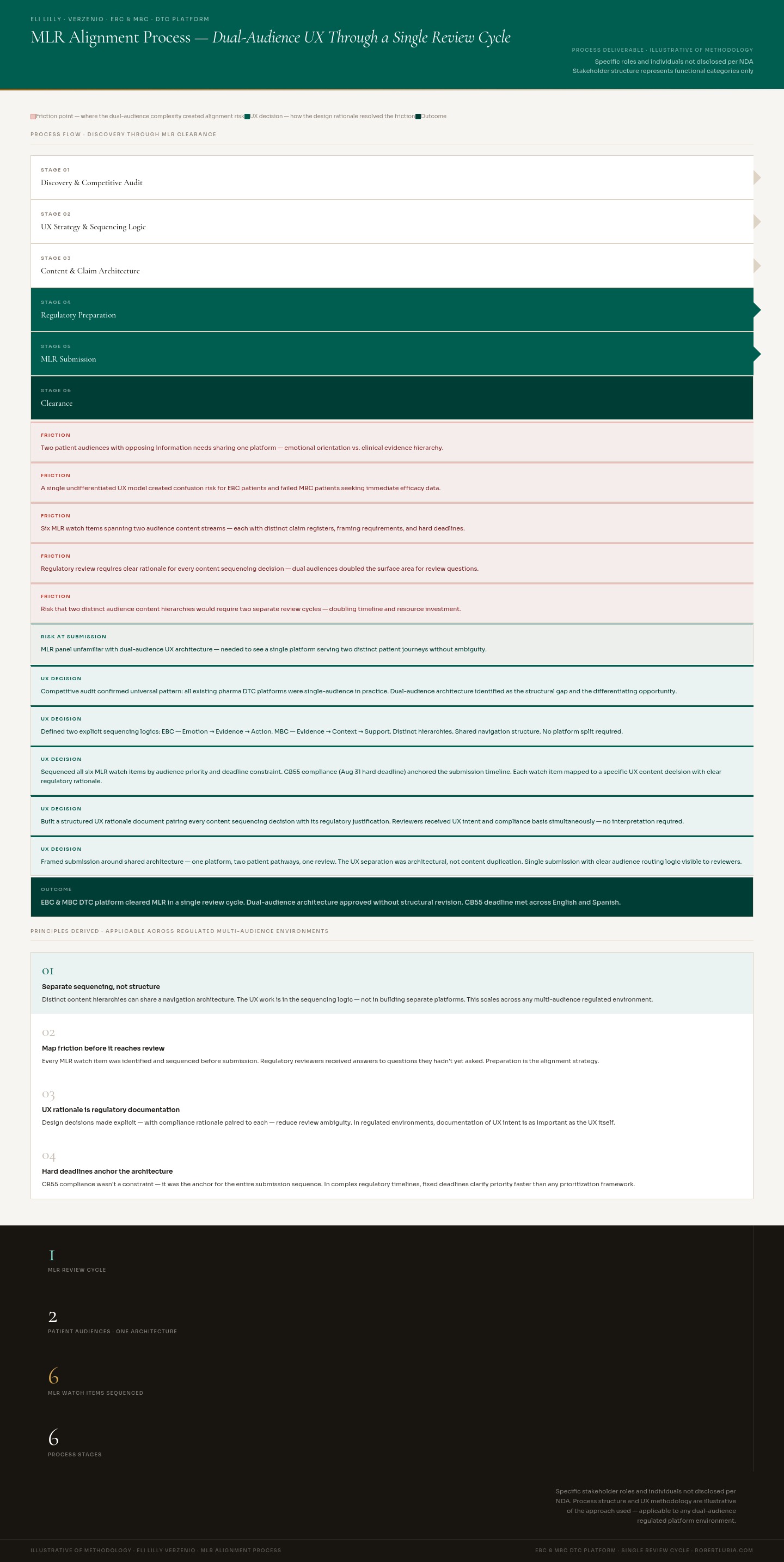
MLR alignment process map — six-stage process showing how a dual-audience UX architecture (EBC and MBC) was brought through a single MLR review cycle. Each stage documents the friction point created by serving two opposing patient needs within one platform, and the UX decision that resolved it. Specific stakeholder roles not disclosed per NDA; process structure illustrative of methodology.
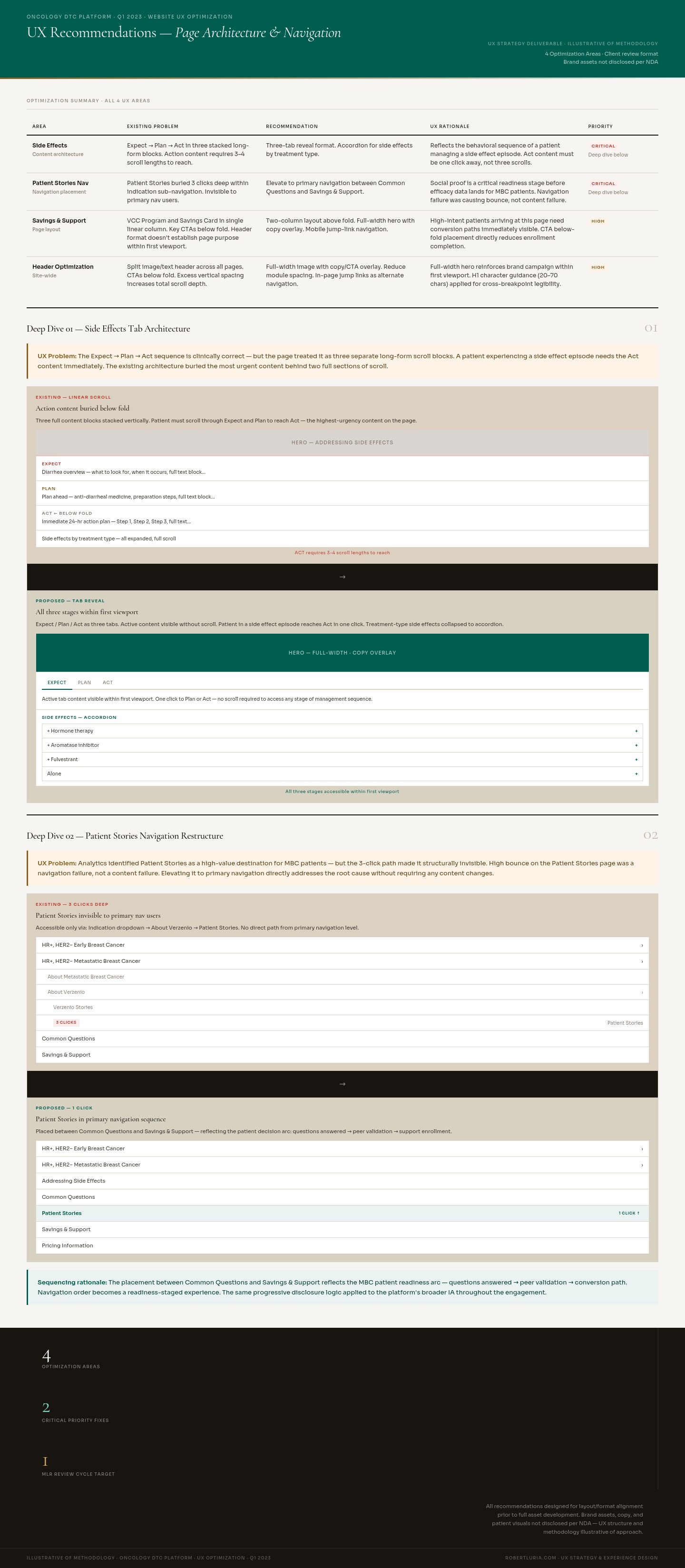
UX optimization recommendations — four page-level UX improvements delivered as a client-facing strategy document covering Savings & Support layout restructure, Side Effects tab architecture (Expect → Plan → Act), Patient Testimonial navigation elevation, and site-wide header optimization. Each recommendation presented as before/after schematic with UX rationale. Specific brand assets not disclosed per NDA; structure and methodology illustrative.

Side Effects page tab architecture — mid-fidelity wireframe comp showing the Expect → Plan → Act tab reveal structure across three states. Left: existing linear scroll layout with action content below fold. Centre: Expect tab active with annotation callouts. Right: Act tab active showing the immediate 24-hour action content surfaced within first viewport. Accordion format for side effects by treatment type shown in lower right. Client review deliverable prior to full asset development.
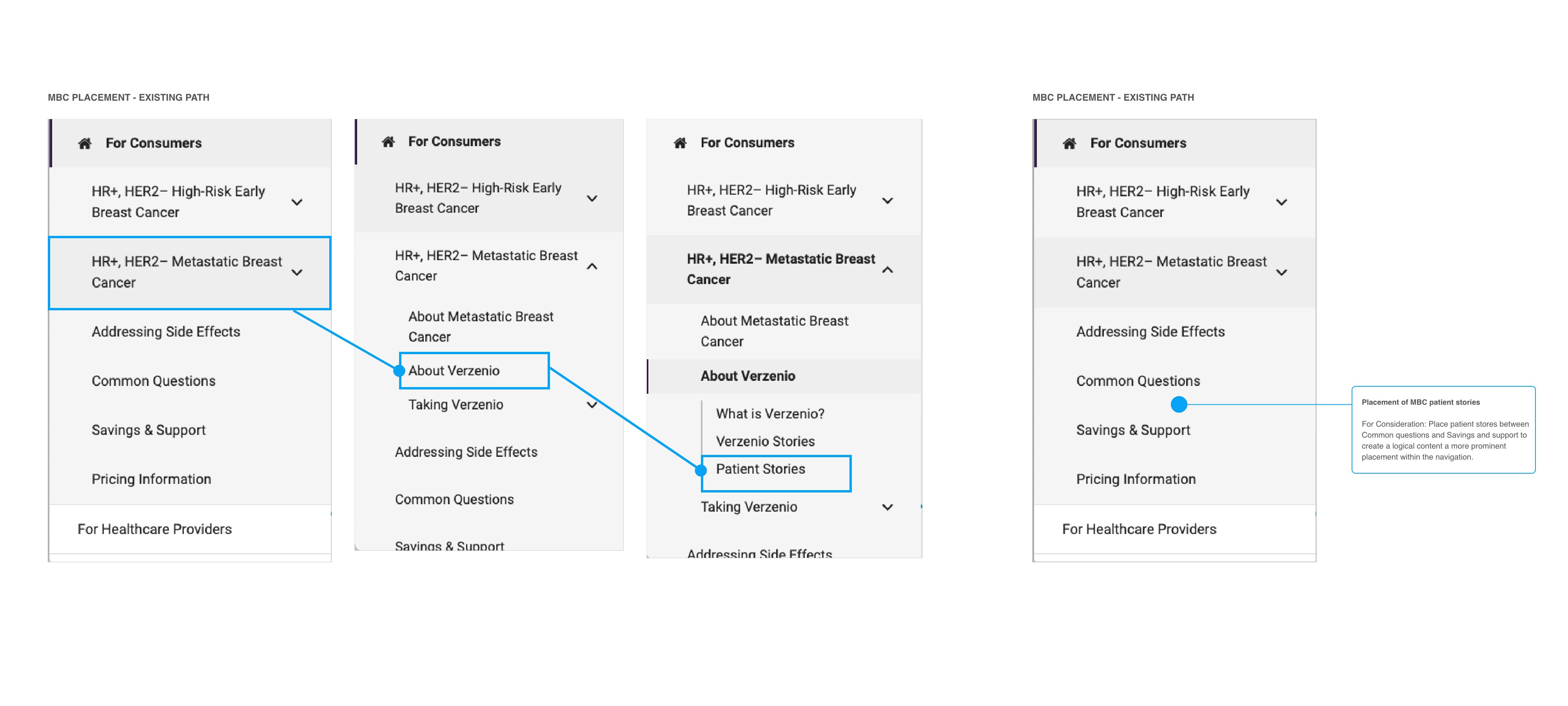
MBC Patient Stories navigation restructure — four-state wireframe showing the existing click path (3 levels deep through indication dropdown → About → Patient Stories) against the proposed primary navigation placement. Blue connector arrows trace the existing buried path. Proposed structure places Patient Stories between Common Questions and Savings & Support — one click from any page, reflecting the patient decision arc from clinical questions to peer validation to support enrollment.
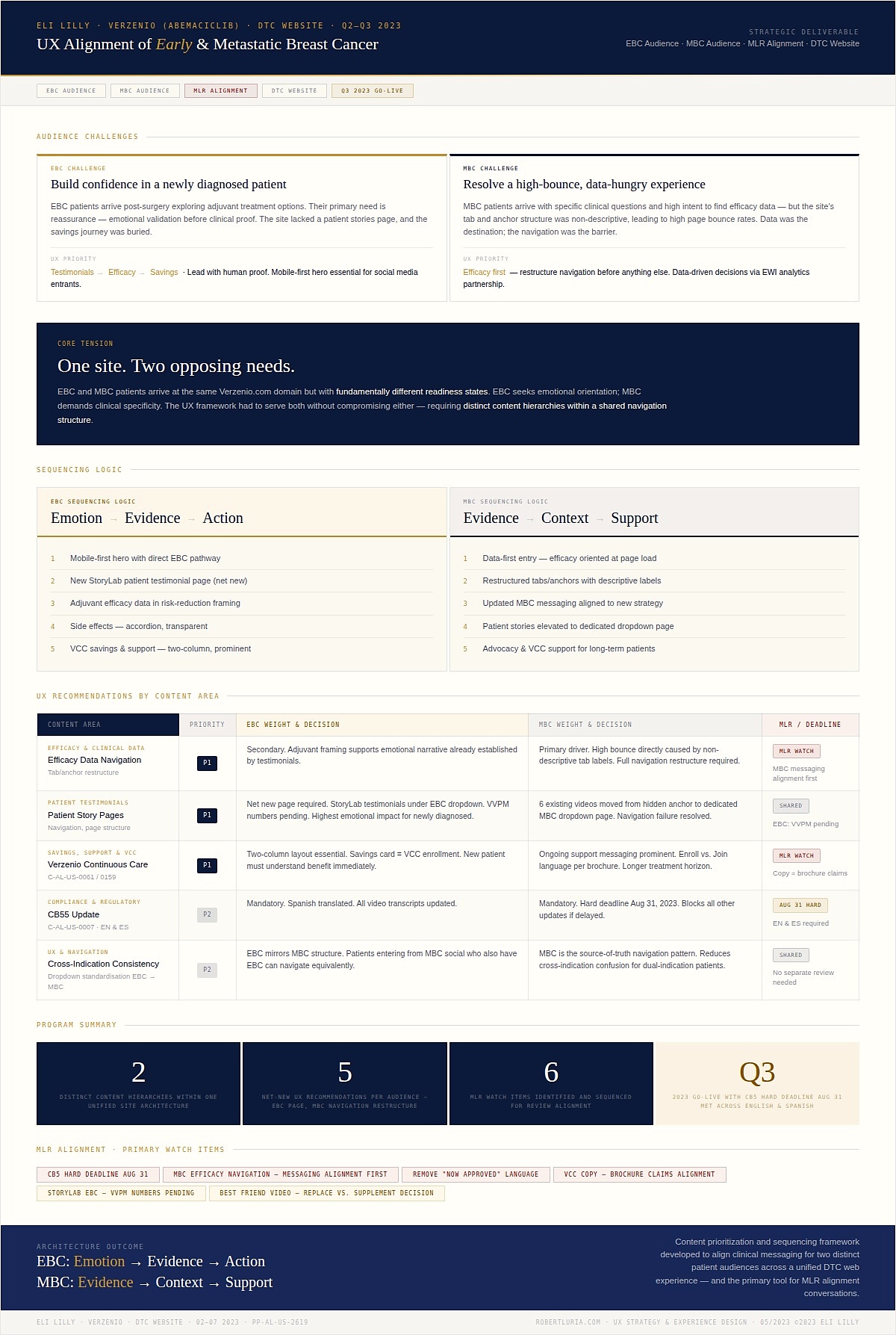
UX alignment framework — the primary UX strategy tool used to align clinical messaging, experience design priorities, and sequencing logic across two distinct patient audiences within a single DTC web experience. Five UX design recommendations per audience across six content areas, with six MLR watch items sequenced for regulatory review alignment.
Mobile UX structural design recommendations — UX structural and navigation improvements across the Verzenio secondary pages for both EBC and MBC patient audiences. Core UX findings identified inconsistent labeling, hero visibility issues, and isolated layouts that increased page length without improving scannability.

MLR submission artifacts — fully produced Verzenio DTC platform delivered following MLR review and approval across both EBC and MBC indications, with distinct visual hierarchy and CTA design for each patient audience following the defined UX sequencing models.
The dual-sequencing architecture was well-supported by the evidence. The one thing I'd shift: build the stakeholder case for structural divergence from the competitive audit outputs — before entering the design phase. When brand, clinical, and MLR teams can see that every competitor is running a single undifferentiated model, the argument for two sequencing architectures lands faster. I'd also bring more MBC patient voice into the evidence validation earlier; the disease-stage literature was strong, but lived experience data would have strengthened the MLR submission case for the Evidence→Context→Support sequence.